Julia Alves Utyama1; Luiz Antônio de Brito Martins2; Guilherme Novoa Colombo Barboza3; Roberta de Júlio Matheus4; Cybelle Guimarães5
DOI: 10.17545/eoftalmo/2019.0007
ABSTRACT
Juvenile primary open-angle glaucoma (JPOAG) is a rare form of glaucoma and is generally detected during childhood or early adulthood. Initially, the affected individuals often exhibit elevated intraocular pressure (IOP) and a significant associated family history. Although there are several factors that influence the clinical management of glaucoma and ocular hypertension, corneal thickness has been a major focus of study in the progression of glaucomatous damage. The association of JPOAG with keratoconus is not yet fully understood; however, several theories have been proposed. This report describes the case of a young female patient diagnosed with keratoconus and with a clinical presentation strongly suggestive of JPOAG. The objective was to analyze the factors involved in these diseases to determine the best methods for the diagnosis and monitoring of such patients.
Keywords: Glaucoma, Open-Angle; Intraocular Pressure; Ocular Hypertension; Cornea; Keratoconus.
RESUMO
O glaucoma primário de ângulo aberto juvenil (GPAAJ) compreende uma forma incomum de glaucoma e geralmente é reconhecido durante a infância ou início da idade adulta. Muitas vezes os indíviduos se apresentam inicialmente com altos valores de pressão intraocular (PIO) e uma forte história familiar associada. Contudo, diante de muitos fatores que podem influenciar no manejo clínico do glaucoma e da hipertensão ocular, a espessura corneana tem sido um dos principais objetivos de estudo para a progressão das lesões glaucomatosas. A associação de GPAAJ com ceratocone ainda não está totalmente elucidada, porém muitas teorias ja foram sugeridas. O presente estudo relata um caso de uma paciente jovem com diagnóstico de ceratocone e curso clínico fortemente sugestivo de GPAAJ. Tem como objetivo analisar os possíveis fatores envolvidos nessas patologias, a fim de encontrar os melhores métodos diagnósticos e acompanhamento desses pacientes.
Palavras-chave: Glaucoma de Ângulo Aberto; Pressão Intraocular; Hipertensão Ocular; Córnea; Ceratocone.
RESUMEN
El glaucoma primario de ángulo abierto juvenil (GPAAJ) es una forma poco frecuente de glaucoma y generalmente es identificado durante la infancia o en el inicio de la edad adulta. Muchas veces los individuos presentan inicialmente altos valores de presión intraocular (PIO) y una fuerte historia familiar asociada. Sin embargo, por encima de muchos factores que pueden influenciar en el manejo clínico del glaucoma y de la hipertensión ocular, el espesor corneano ha sido uno de los principales objetivos de estudio para la progresión de las lesiones glaucomatosas. La asociación de GPAAJ con el queratocono aún no está totalmente determinada, sin embargo, ya fueron sugeridas muchas teorías. El presente estudio describe un caso de una paciente joven con diagnóstico de queratocono y curso clínico fuertemente indicador de GPAAJ. Tiene como objetivo analizar los posibles factores involucrados en esas patologías, a fin de encontrar los mejores métodos diagnósticos y acompañamiento para dichos pacientes.
Palabras-clave: Glaucoma de Ángulo Abierto; Presión Intraocular; Hipertensión Ocular; Córnea; Queratocono.
INTRODUCTION
Keratoconus is a bilateral, asymmetric, progressive, and degenerative corneal disease; it is characterized by the central thinning of the cornea with apical protrusion and irregular astigmatism. The disease commonly manifests during late adolescence or early adulthood, and despite the associated factors, its etiology remains unclear1. Previous studies suggest that its physiopathogenesis and progression are the result of several physical, biochemical, and molecular factors2.
Glaucoma is an optic neuropathy of multifactorial origin, characterized by progressive damage to the optic nerve and subsequent visual field loss3. It is estimated that glaucoma affects more than 66 million individuals worldwide; among these, up to 6.8 million individuals suffer from blindness. Juvenile primary open-angle glaucoma (JPOAG) is an uncommon form of glaucoma. It is generally detected during childhood or early adulthood and is associated with a significant family history4. The biological mechanisms underlying this disease are still not fully understood and the factors that contribute to its progression remain unclear. However, elevated IOP is currently the only demonstrated and treatable risk factor5. Previous studies indicate that changes in corneal structure lead to altered IOP values, particularly when measured by Goldmann applanation tonometry (GAT), the gold standard method. Therefore, the cornea and pachymetry have become the objects of study in glaucomatous damage6. Central corneal thickness (CCT) has been shown to be the most important risk factor in patients with ocular hypertension. This is because IOP values measured using GAT are underestimated in thinner corneas and overestimated in thicker corneas, which delays diagnosis and undermines glaucoma monitoring in such patients7,8.
Here, we aimed to describe the case of a patient with keratoconus and a clinical presentation suggestive of JPOAG, and analyze the possible pathophysiological factors underlying the association between these diseases, to contribute to early diagnosis, control of IOP, and adequate glaucoma monitoring. An additional purpose was to demonstrate the importance of investigating glaucoma in patients with keratoconus because it may hinder a timely diagnosis and even be correlated with the development and progression of the disease.
CASE REPORT
A 28-year-old woman, a shopkeeper, presented with complaints of low visual acuity (VA) that persisted for more than one year. She reported not being able to wear correction contact lenses (CL) and that wearing glasses did not improve her eyesight. She had been using latanoprost once a day for the previous six months, prescribed at another healthcare center. She denied having any eye or systemic diseases. She had a family history of primary open-angle glaucoma and advanced JPOAG, including first-degree relatives. Upon examination, corrected VA (CVA) was 0.80 and 0.60. Biomicroscopy and gonioscopy did not reveal any changes. IOP was 12 and 20 mmHg and the optic disc examination showed normal optic cup-to-disc ratio. Topography, pachymetry, retinography (optic disc photograph), and visual field exams were requested.
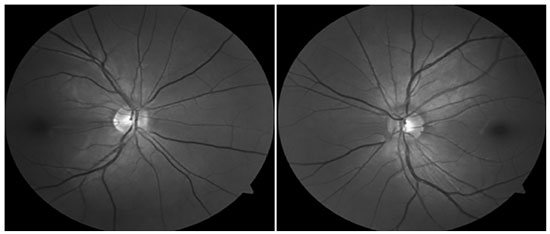
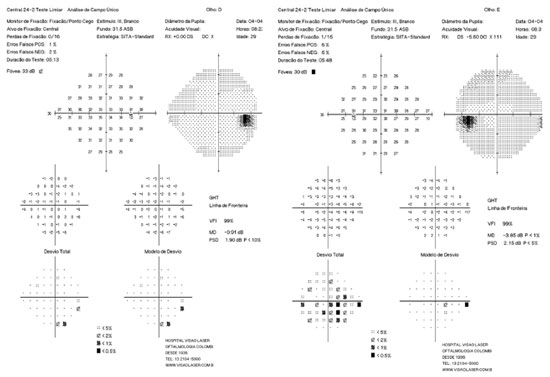
Mean keratometry was 46.70 and 50.62 D, central pachymetry was 478 and 463 mm, and topography findings were compatible with keratoconus according to the Rabinowitz criteria (Figure 1). Retinography did not show changes suggestive of glaucoma (Figure 2) and the visual field test showed widespread decrease in sensitivity, more pronounced on the left eye (OS) (Figure 3). Corneal rigid CL fitting was indicated, to which the patient adapted well, and VA improved to 1.0 and 0.8; moreover, medication washout was indicated to evaluate basal IOP, which increased to 40 and 43 mmHg (morning values) within one month. Latanoprost was resumed and administered once a day. Subsequently, optical coherence tomography of the optic nerve and macula showed a mean thickness of the nerve fiber layer within normal values, in both eyes (OU).
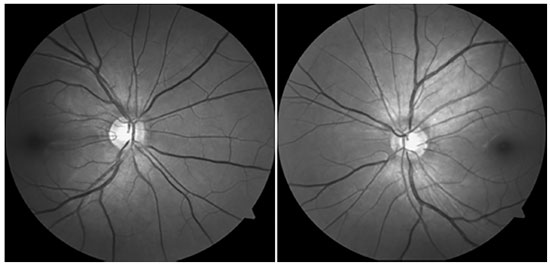
After 18 months of follow-up, although the patient’s IOP was under control (13 and 15 mmHg), she started exhibiting changes suggestive of glaucomatous progression. Examination of the ocular fundus showed slight increase in the cup-to-disc ratio (Figure 4) and visual field with initial inferior nasal step (OS) (Figure 5). In a second pachymetry exam, CCT was 478 and 456 mm. In the subsequent appointments, IOP was to be increased to 36 mmHg (OU). The drugs she was taking were then replaced by combinatorial therapy with brimatoprost and timolol. The patient is currently being monitored and her IOP is under control (14 and 16 mmHg). She has adapted well to rigid CL, has a CVA of 0.8 OU, and has no complaints.
DISCUSSION
Glaucoma and keratoconus may be interlinked in several ways. The prevalence of keratoconus among patients with glaucoma and in the general population varies between 0.7% and 3.3% and between 0.05% and 2.3%, respectively9,10.
According to Ambrósio, the mean CCT in the population is 556 ± 35 mm, and the article of the Ocular Hypertension Treatment Study (OHTS) in 2002, revealed that the CCT value is an important predictive factor of glaucoma development in patients with ocular hypertension6. Several theories explain this fact. Corneas with a thickness outside of the normal limits may cause errors in the measurement of IOP and, therefore, in its interpretation. In the case of keratoconus, the decrease in CCT may lead to an underestimation of the results and hinder the diagnosis, monitoring, and treatment of glaucoma patients. Other studies suggest that glaucomatous damage progression may be related not only to IOP but also to ocular tissue compliance. Corneal hysteresis may be related to the biomechanical properties of the cribriform plate and subsequent exposure of the optic nerve11,12. This theory is consistent with the association between the axial length of the eye, myopia, and its higher susceptibility to glaucoma, already reported in some studies13,14. It is also based on the fact that, even with adequate control of IOP, glaucomatous damage may continue to progress15.
To address the difficulties in managing the IOP of patients with corneal disease, several studies suggest that although the role of corneal hysteresis in IOP and glaucoma progression has not yet been fully clarified, it is important to use complementary methods for the biomechanical and tomographic evaluation of the cornea. Many of these authors believe that the use of technologies such as the ocular response analyzer and Pentacam provides additional information about the condition of the cornea and benefits the understanding of IOP and the prognosis of such patients6,11,16.
Another challenge in the management of patients with glaucoma and keratoconus is good quality imaging in the evaluation of the optic nerve. The suspected nerves may be considered normal, not only because IOP values may be underestimated but also because of the severe myopia that the patient usually exhibits. This occurs as a result of irregular astigmatism and scarring of these corneas, which limit the assessment of glaucoma4,15.
JPOAG is a rare form of glaucoma, characterized by high levels of IOP and progressive damage to the optic nerve and nerve fiber layer. Similar to other forms of glaucoma, it requires careful investigation and understanding to prevent an entire life without eyesight17.
The underestimation of IOP, the potential biomechanical changes in the cornea, and the limited quality of the images obtained in the optic disc examination when evaluating individuals with keratoconus may substantially contribute to the establishment and progression of glaucoma.
In the treatment and monitoring of the reported clinical case, it is essential to consider the interferences caused by an abnormally thin cornea. Thus, considering the potentially underestimated IOP values, the need to achieve a thorough control of IOP is unquestionable. Although there are no standard protocols for the treatment and monitoring of such patients, they should also undergo regular and frequent testing of the visual field and optic nerve, even when these were initially normal.
In conclusion, this study indicates the need for awareness regarding the coexistence of JPOAG and keratoconus, so that clinical decisions are always made by taking into consideration all the pathophysiological factors that potentially underlie the association between these two conditions.
REFERENCES
1. Elias RMS, Lipener C, Uras R, Pavês L. Ceratocone: fatores prognósticos. São Paulo: Arq Bras Oftalmol. 2005 July/Aug; v. 68, n. 4.
2. Lopes ACN, Pinto AGT, Sousa BA. Ceratocone: uma revisão. Brasília: Rev Med Saúde. 2015; 4(2):219-232.
3. Urbano AP, Freitas TG, Arcieri ES, Urbano AP, Costa VP. Avaliação dos tipos de glaucoma no serviço de oftalmologia da UNICAMP. Arq Bras Oftalmol. 2003; 66:61-65.
4. Sahil MBBSG, Anita MDG, Viney MDG. Bilateral juvenile onset primary open-angle glaucoma among keratoconus patients. J Glaucoma. 2015 April/ May; 24(4):e25-e27.
5. Weinreb RN, Khaw PT. Primary open-angle glaucoma. Lancet, 2004 May; 363:1711-1720.
6. Silva JA, Silva RS, Ambrósio Jr R. Relevância da biomecânica da córnea no glaucoma. Rev Bras Oftalmol. 2014; 73(1):37-39.
7. Gordon MO, Beiser JA, Brandt JD, Heuer DK, Higginbotham EJ, Johnson CA, et al. The Ocular Hypertension Treatment Study: baseline factors that predict the onset of primary open-angle glaucoma: Arch Ophthalmol. 2002; 120(6):714-720, discussion 829-830.
8. Gordon MO, Torri V, Miglior S, Beiser JA, Floriani I, Miller JP, et al. Validated prediction model for the development of primary open-angle glaucoma in individuals with ocular hypertension. Ophthalmology. 2007; 114(1):10-19.
9. Jonas J, Nangia V, Matin A, et al. Prevalence and associations of keratoconus in rural Maharashtra in central India: the Central India Eye and Medical Study. Am J Ophthalmol. 2009; 148:760-765.
10. Rabinowitz YS. Keratoconus. Surv Ophthalmol.1998; 42:297-319.
11. Congdon NG, Broman AT, Bandeen-Roche K, Grover V, Quigley HA. Central corneal thickness and corneal hysteresis associated with glaucoma damage. Am J Ophthalmol. 2006; 141:868-875.
12. Brown KE, Congdon NG. Corneal structure and biomechanics: impact of the diagnosis and management of glaucoma. Curr Opin Ophthalmol. 2006; 17:338-343.
13. Mitchell P, Hourihan F, Sandbach J, Wang JJ. The relationship between glaucoma and myopia: The Blue Mountains Eye Study: Ophthalmology. 1999; 106:2010-2015.
14. Wong TY, Klein BE, Klein R, Knudtson M, Lee KE. Refractive errors, intraocular pressure, and glaucoma in a white population. Ophthalmology. 2003; 110:211-217.
15. Elisabeth BYJ, Cohen MD. Keratoconus and normal-tension glaucoma: a study of the possible association with abnormal biomechanical properties as measured by corneal hysteresis (an aos thesis): Trans Am Ophthalmol. 2009 Dec; 107:282-299.
16. Perucho GL, de la Casa JMM, Morales FL, Baneros RP, Saenz FF, Garcia FJ. Intraocular pressure and biomechanical corneal properties measure by ocular response analyser in patients with primary congenital glaucoma. Acta Ophthalmol. 2016 Aug; 94(5):e293-297.
17. Thau A, Lloyd M, Freedman S, Beck A, Grajewski A, Levin AV. New classification system for pediatric glaucoma: implications for clinical care and a research registry. Curr Opin Ophthalmol. 2018 Sep; 29(5):385-394.





Funding: No specific financial support was available for this study
Disclosure of potential conflicts of interest: The authors have no potential conflicts of interest to disclose
Received on:
October 5, 2018.
Accepted on:
February 18, 2019.